Description
IRIS Salmonella constitue une méthode alternative de recherche des salmonelles dans les produits d’alimentation humaine et animale, ainsi que dans les échantillons de l’environnement (à l’exclusion des échantillons de production primaire).
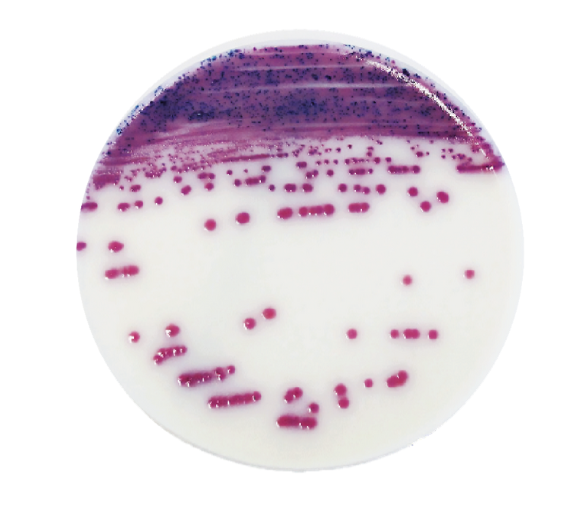
Les études réalisées avec IRIS Salmonella Agar démontrent sa grande spécificité pour la détection des salmonelles, y compris les espèces et les sérovars atypiques, sources de confusions sur d’autres milieux.
Ainsi, la mise en évidence des Salmonella Typhi et Paratyphi, des Salmonella lactose-positif (Salmonella Senftenberg et sous-espèces S. arizonae et S. diarizonae), des souches saccharose-positif est assurée. Le milieu permet la détection des sérovars immobiles (S. Pullorum et S. Gallinarum) ou des souches monophasiques. IRIS Salmonella Agar permet également de détecter les souches présentant une activité estérasique faible à nulle sur d’autres milieux (Salmonella bongori, Salmonella Dublin et Atento, quelques unes des sous-espèces S. enterica S. houtenae et S. diarizonae).
IRIS Salmonella est certifiée NF VALIDATION, sous Attestation N° BKR 23/07 – 10/11, pour tous les produits d’alimentation humaine et animale, ainsi que les échantillons de l’environnement (hors échantillons d’environnement de production primaire).
La méthode est également validée pour les prises d’essai de 50 g à 375 g pour les poudres de lait (incluant les poudres de lait infantile avec et sans probiotiques) et les prises d’essai de 50 g à 125 g pour les farines et les croquettes utilisées dans l’alimentation animale.
Conditionnement
IRIS Salmonella Agar :
BM16008 – 20 boîtes de Petri (Ø 90 mm)
BM16108 – 120 boîtes de Petri (Ø 90 mm)
Salmonella Enrichissement :
BK194HA – Flacon de 500 g
BK194GC – Fût de 5 kg
BM13608 – 10 flacons de 225 mLBM14408
BM13708 – 3 poches souples de 3 litres
BM14408 – 2 poches souples de 5 litres
BM23708 – 40 poches souples de 5 litres
Salmonella Enrichissement + Tween® 80 :
BM16308 – 3 poches souples de 3 L
BM19808 – 2 poches souples de 5 L
BM22808 – 10 flacons de 225 mL
IRIS Salmonella Supplément :
BS07808 – 10 flacons de 50 mL
BS07708 – 120 comprimés qsp 225 mL
BS09308 – 120 comprimés qsp 90 mL
IRIS Salmonella Supplément concentré :
BS10208 – 10 flacons de 90 mL
Kit d’agglutination au Latex :
BT01108 – CONFIRM’ Salmonella
Documentation
- FDS_MSDS_BK212_v1.pdf
- FDS_MSDS_BS07708_v1.4
- FDS_MSDS_BS07808_v1.1.pdf
- FDS_MSDS_BS09308_v1.3.pdf
- FDS_MSDS_BS10208_V1.1.pdf
- FT_IRIS SALMONELLA_BM16008_BM16108_BS07708_BS09308_BS07808_FR_v9.pdf
- Non classification statement_BM13608_BM13708_BM14408.pdf
- Non classification statement_BM16008_BM16108
- Non classification statement_BM19808.pdf
- Non classification statement_BM22808.pdf


